Lab-grown diamonds have the same carbon structure as naturally occurring ones. But while the former is made over millions of years, the synthetic kind are formed in days or weeks.
They’re generally grown using a high-pressure, high-temperature (HPHT) method. Scientists at General Electric designed the first HPHT method in 1954 to mimic diamond growth deep below the Earth’s surface. HPHT is the most economical process for growing large diamonds.
1. Sow the seed

The HPHT process begins with a tiny diamond fragment, known as a seed crystal. This is placed in a cell inside a press, together with a metal catalyst, such as nickel or iron. A block of pure carbon, such as graphite, sits on top.
2. Apply pressure

The press heats the cell to a searingly high temperature, up to 1,600°C. It then uses metal anvils to apply intense pressure. The pressure and temperature need to be high enough to melt the metal catalyst.
3. Atoms accumulate
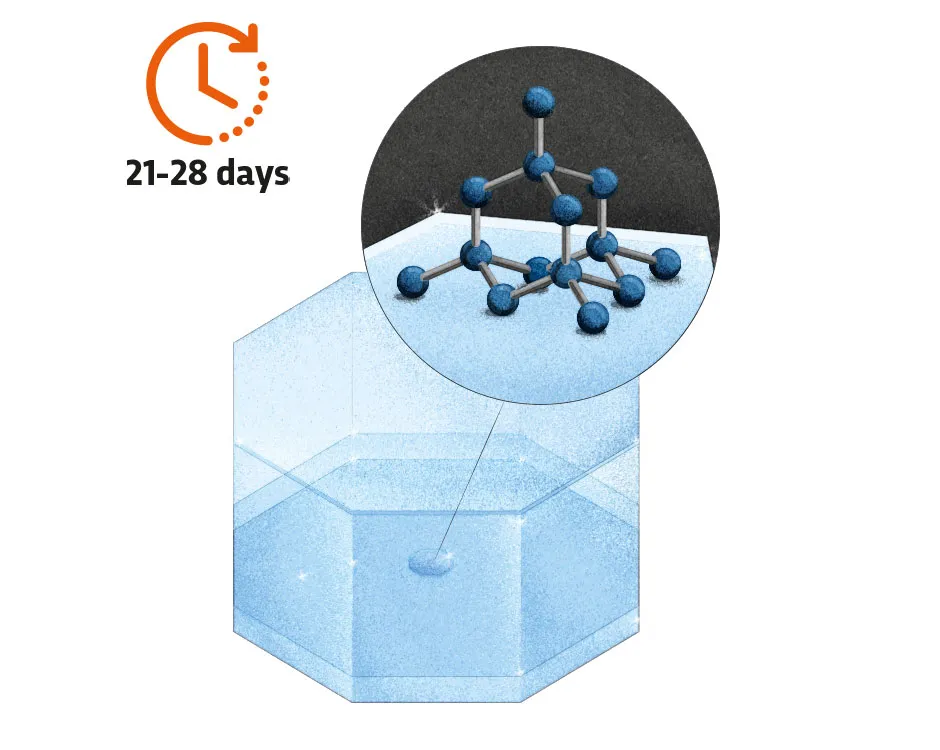
The molten metal carries carbon atoms from the graphite to the seed crystal. The carbon atoms slowly build up around the crystal to give the multi-sided structures that we know so well. The growth rate needs to be controlled to reduce the risk of faults.
4. Cut and polish

After the diamond has been cooled, it’s ready to be cut and polished, just like nature’s finest. It can then be graded for cut, colour, carat and clarity. The catalysts can give the diamonds a touch of colour, depending on the type of metal.
Read more: