Sitting on the top tier of a cruise ship in Egypt, Professor Steffanie Strathdee and her husband were enjoying their meal and looking forward to sightseeing on their final day. It had been a dream holiday for Steffanie's husband, Tom, to explore Egypt and the pair of researchers had finally made it over in 2015. A few hours after their idyllic dinner, however, Tom suddenly fell terribly ill.
Professor Steffanie Strathdee, an infectious disease epidemiologist, supposed it was from the seafood they’d eaten and didn’t think much of it until the next day. Nothing worse than food poisoning ruining your final day on holiday, right? Well, as he became more dehydrated and other symptoms started to crop up, Steffanie began to worry that something worse had disrupted their bliss.
“I'm not a medical doctor, but I do have a rusty old degree in microbiology. And I was literally calculating incubation periods in my head for different organisms that he could have acquired. When he started complaining of back pain, I realised this isn't food poisoning if he's got back pain. So I called a friend of ours who happened to be the head of infectious diseases in the department we work,” says Steffanie.
This friend was Professor Chip Schooley at the University of California, San Diego. He played an essential role in unravelling this mystery illness. From the symptoms described, he urged Steffanie to take Tom to a hospital as soon as possible. Unfortunately, the area of Egypt they were staying in didn’t have any hospitals, only a clinic.
“[The clinic] was staffed with wonderful Egyptian doctors and they did their best with what limited resources they had,” Steffanie says. “They did diagnose pancreatitis, an inflammation of the pancreas but they said there would likely be some serious complications that are going to be above what the clinic can handle.”
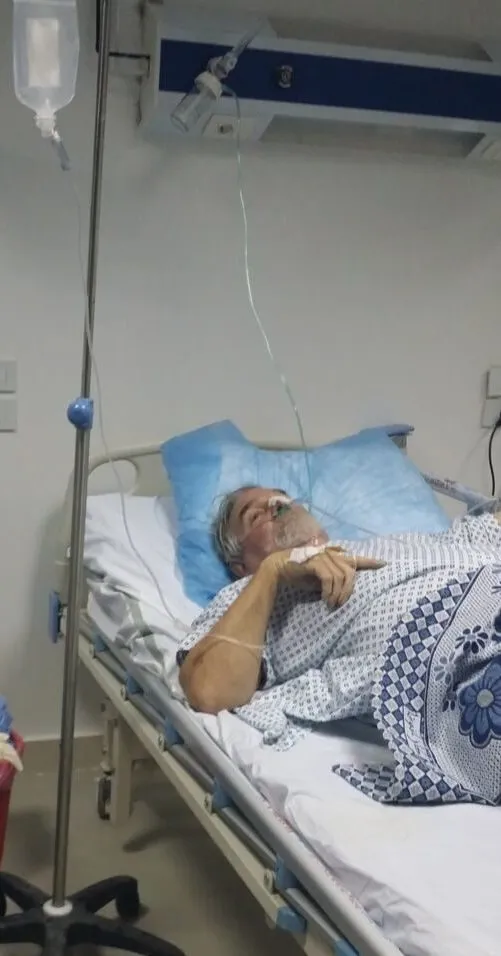
So, Steffanie, her husband and his inflamed pancreas travelled over 3,000km from Egypt to reach a hospital in Germany. Once in the hospital, shewas shocked to learn that her husband had a gallstone obstructing his bile duct that had led to a stomach abscess “the size of a small football.”
Draining the abscess revealed a putrid fluid. There was something growing in there, the doctor said, and it had been for some time.
Staring down a flask of this murky stomach fluid, Steffanie wasn’t too alarmed by the doctor’s news. She knew that generally these things can be resolved by examining the material that had been growing in the abscess and prescribing the relevant antibiotics. A fairly straightforward problem to tackle. “He’ll be right as rain in a couple of days,” she thought.
Read more about medicine and antibiotics:
- The history of medicinal drugs helps explain our relationship with them today
- How do antibiotics work?
- Should medicine be gendered
Unfortunately, this wasn’t the case for Tom. A few days passed and rather than arriving with good news, the doctor returned in head to toe protective medical clothing. Standing aside from Tom’s bed with a gown, mask and a set of gloves he announced to Steffanie, “I’ve got some very bad news. This is the worst bacteria on the planet.”
What makes a bacteria good or bad in a case like this is how treatable it is. Can we throw penicillin at it and solve everything? Or does it need more tailored antibiotic treatment? If the bacteria causing your illness adapts to dodge the antibiotics trying to clear your body, then it is very bad indeed.
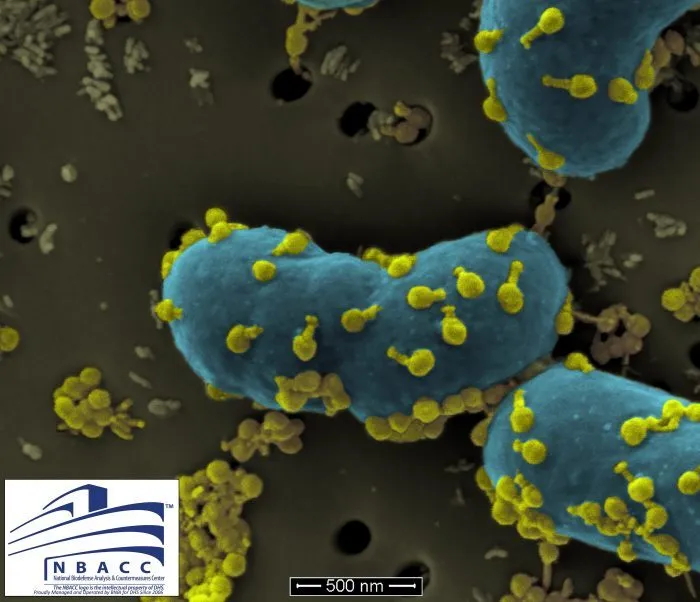
Antibiotic resistant bacteria which persist despite antibiotic treatments can be fatal. This bacteria, Acinetobacter baumannii,may not be familiar to most, but Steffanie recognized it from her undergraduate microbiology degree in the 1980s.
“I realised that we used to plate this organism on our petri dishes back then and all we needed was a lab coat and gloves,” she says. “And he said this is the worst bacteria on the planet. Like, how can this be? I'm an infectious disease epidemiologist. This is catching me really off guard.”
The worst bacteria in the world
Native to soil and water bodies across the world, Acinetobacter baumanniiwas considered to be sensitive to most antibiotics in the 1970s and 80s. However, today it shows wide-ranging resistance to several antibiotics and is regarded as a superbug. This bacteriacolonises the body, specifically targeting moist tissue and causing necrosis (tissue cell death) before entering the blood stream.
Steffanie investigated and learned how the bacteria rapidly develops resistance through genetic mutations, eschewing treatment. “It’s something of a bacterial kleptomaniac. It’s really good at stealing antibiotic resistant genes from other bacteria,” says Steffanie. Though concerned, she knew the doctors would try the options available to defeat the superbug and improve Tom’s condition.
The doctors in Germany tried a range of antibiotic treatments for Tom. While the Acinetobacter baumanniifestered and developed, these treatments effectively wiped out his immune system and allowed the superbug to spread.
Steffanie says that while in Germany, the superbug was resistant to 15 of the antibiotics they tried and was only partially sensitive to three. However, all of these three had severe side effects and is used as a last resort antibiotic. They decided it was time to move Tom home to San Diego, taking care to follow infection control procedure to prevent his condition from declining.
By the time they arrived back in San Diego, Tom’s superbug had become resistant to these last three antibiotics. At this point, Steffanie relied on her colleagues and the doctors for the best course of treatment. He was at risk of septic shock, a potentially fatal condition in which your blood pressure plummets to a dangerously low level. So, the doctors in San Diego tried to siphon the infected fluid from his abscess as Tom was too weak for surgery. Yet, the day before they were supposed to leave hospital, the superbug spread further.
“He had five different drains as well as a feeding tube, he looked like a pincushion. One of those internal drains slipped and it poured all that infected fluid into his abdomen, into his bloodstream,” recalls Steffanie.
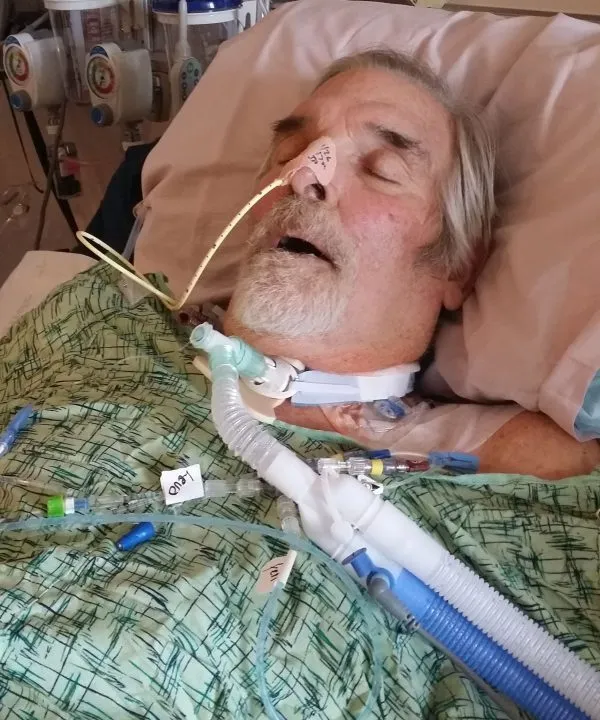
This spillage resulted in what doctors had been dreading. Tom went into septic shock.
“Your body is kind of over reacting to an invader that hits the bloodstream. And right away, the heart rate increases, the blood pressure drops, the person flushes, develops a fever and starts panting and usually they start to shake. This shaking is called rigors and it's so intense that, in Tom's case, the bed frame hit the wall. And this is all within like the span of two minutes, so I was pretty stunned, ” says Steffanie.
Spreading superbug and searching for solutions
He was rushed to intensive care where doctors put him on a ventilator. Steffanie had thought she would be taking her husband home the following day, but instead Tom ended up in a medically induced coma. After reaching his bloodstream, the superbug fully colonised Tom’s body.
At this point, with no antibiotics left, Steffanie began to investigate further, researching the superbug and alternative treatment possibilities. Reflecting on this now she says, “it hadn't really sunk in that I might never be able to hear my husband speak again. We may never be able to communicate.
“There was nothing else the doctors could do and little by little he was wasting away. After some time, he was not in a medically induced coma. He was in a coma that he wasn’t really waking up from. Some days his eyebrows would kind of wiggle or his fingers would try to reach out,” she says.
With Tom’s lungs failing, medications keeping his heart pumping and doctors suggesting he needed kidney dialysis, Steffanie knew it was close to the end. Sitting by his bedside, she asked if he wants her to keep fighting for him.
“I know that you're fighting really hard and you're tired. And if you want to let go, I'll understand. But I want to grow old with you. And if you want to live, please squeeze my hand and I'll try to figure out something to do to stop this thing. And I waited and he squeezed my hand,” she shares.
Though elated, she was now presented with a seemingly insurmountable challenge: “I realised I’m not a medical doctor, what am I going to do? I am a scientist and I know how to research,” Steffanie says.
“So, I went home and I hit the internet and the National Library of Medicine has a free search engine that is like Google Scholar on steroids. It's open to the public, so even your grandmother can plunk in keywords like the name of a superbug and alternative treatments and up pops scientific literature that's been vetted and is based on real evidence,” Steffanie says.
As an epidemiologist focusing on the HIV epidemic, the superbug crisis crept out from the shadows the more she researched. Buried in the literature she found her solution: phage therapy.
Read more about antibiotic resistance:
- What causes antibiotic resistance?
- Will the rise of superbugs return us to a world without antibiotics?
- Antibiotic resistance: Is it really as bad as climate change?
Phage therapy
Though still regarded as an experimental treatment in the Western world, phage therapy was first used to treat bacterial infections in 1919. It involves deploying bacteriophages (bacteria-eating organisms) which can target and hijack bacteria cells to reproduce and kill the host cells.
With the rise of penicillin following WWII and subsequent geopolitical influences, phage therapy has been under-researched in the West. As a result, Steffanie had to convince her family that this experimental treatment was the right thing to do to.
After this she still faced series of obstacles. First, she had to find experts in the field willing to help her husband, and then they had to identify and purify the bacteriophages for treatment. To treat Tom, doctors would first need FDA approval. Schooley, who had first recommended taking Tom to a hospital, offered to help get FDA permission for bacteriophage treatment on a compassionate basis.
“I was very excited again, but I was daunted by this seemingly impossible task because the more I learnt about phages, the stranger the whole idea got. Essentially, phage are the oldest and most populous organism on the planet. There's 10 million trillion trillion - and they have to match to the bacteria that you want to kill,” says Steffanie.
Phages use specific receptors to attack the superbug, so she couldn’t just use the most common phage for Tom. Steffanie now had to tackle the problem of finding the specific strain of bacteriophage that could save her husband.
Finding phages
Determined to find a solution, Steffanie emailed the few researchers she could find working on Acinetobacter baumanniiand the associated phages. Attaching a picture of Tom in his hospital bed, she reached out to the scientists in the field for help. Within 24 hours, she says she heard back from Professor Rylan Youngat A&M Texas University who reached out to others internationally. Young also refocussed his lab on looking for these phages from waste and sewage sources.
Isolating four relevant bacteriophages from sewage waste, the Texas lab then let it grow and tested it on a sample of the bacteria. After successful testing in a petri dish, the team had to purify their bacteriophages for human use.
In just a few weeks, Steffanie had a set of phages from Texas ready for use. The US Navy also provided a different set of active phages against Acinetobacter baumannii.
With permission for treatment on an emergency basis thanks to Professor Schooley, Steffanie had overcome an almost impossible task. Hours of research, discussion with doctors, contacting experts, and collaboration with labs alongside a hand-squeeze of hope had led Steffanie to a potential cure for Tom.
From sewage to treatment
Usually phage therapy involves topical treatment, spreading the bacteriophages on the skin. But, with Tom’s dire condition, Schooley had scrutinised the research and, after speaking to experts, he recommended intravenously administering the phages. At the time, IV phage therapy had never been attempted in the US and they had to guess the dosage for Tom.
“When we injected the phages into Tom's bloodstream, nobody really knew if this was going to cure him or kill him. We did know that if we didn't do something, that he was going to die anyway. I was told that they expected him to die within a couple of hours,” says Steffanie.
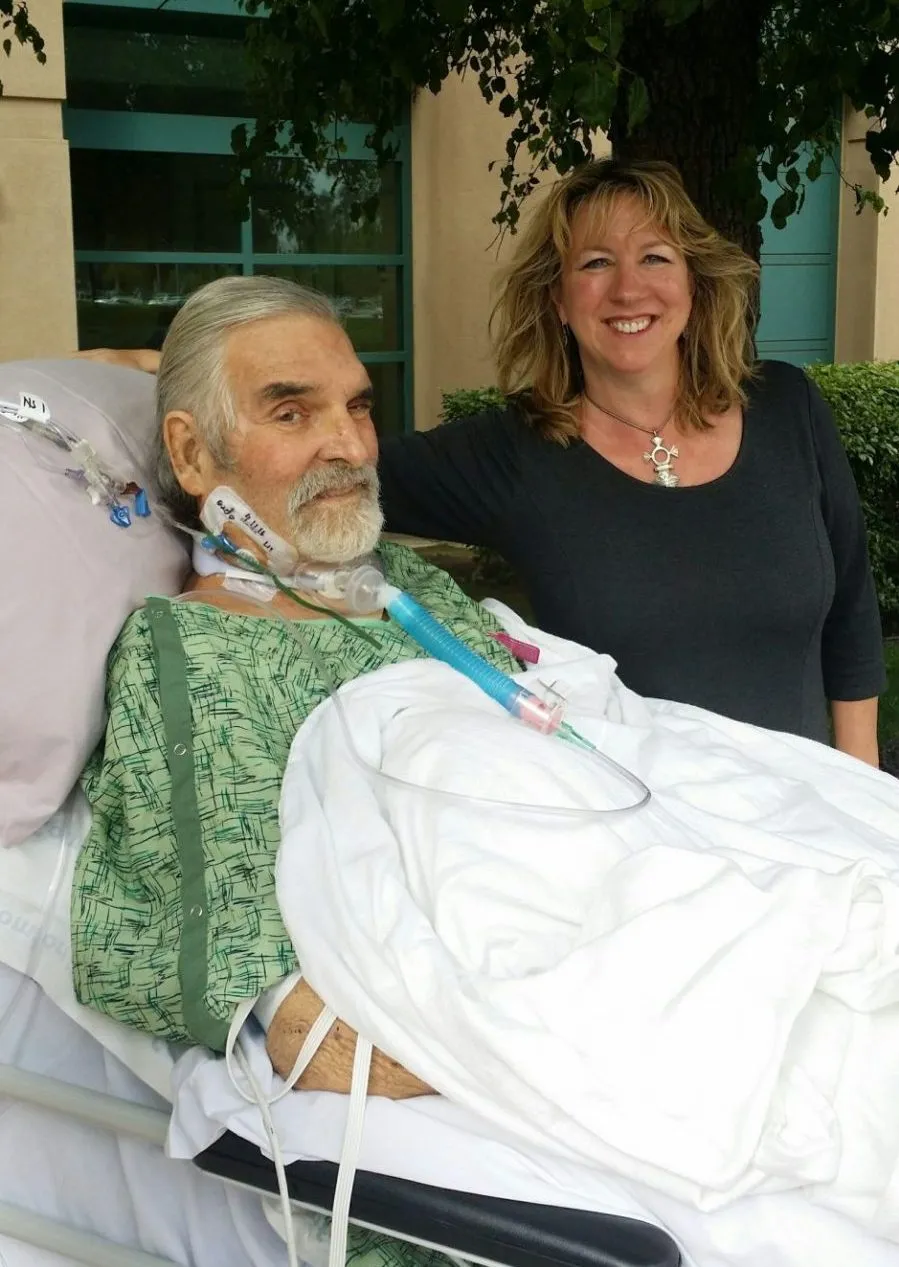
“And yet, you know, three days after we started injecting the phage into his bloodstream, he lifted his head off the pillow, opened his eyes and kissed his daughter's hand. It was just fantastic.”
Steffanie says Tom has made an incredible recovery, and cleared the infection in three months. He can take long walks every day. “He even takes out the garbage every time I ask,” she says.
Learning microbiology in 1986, Steffanie could have never envisioned her lectures on bacteriophages would play such a role in her personal battle years later. Her work to save Tom’s life helped renew interest and research into phage therapy in the West which has treated others since.
Professor Steffanie now also directs a centre in San Diego focused on developing phage therapy for medicine. Research into phages and other therapeutic interventions will be key to overcoming the antibiotic resistance crisis.
Together, the pair published a book in 2019 about their experience and the potential of phage therapy,The Perfect Predator. Not quite the dream ending to their vacation in Egypt, but certainly a holiday story to trump all others.
Listen to Steffanie tell the story on theScience Focus Podcast:
The Perfect Predator: A Scientist's Race to Save Her Husband from a Deadly Superbug by Steffanie Strathdee and Thomas Patterson is out now (£14.99, Hachette Books).
- Buy now from Amazon UK, Waterstones or WHSmith


