1
Salmonellae
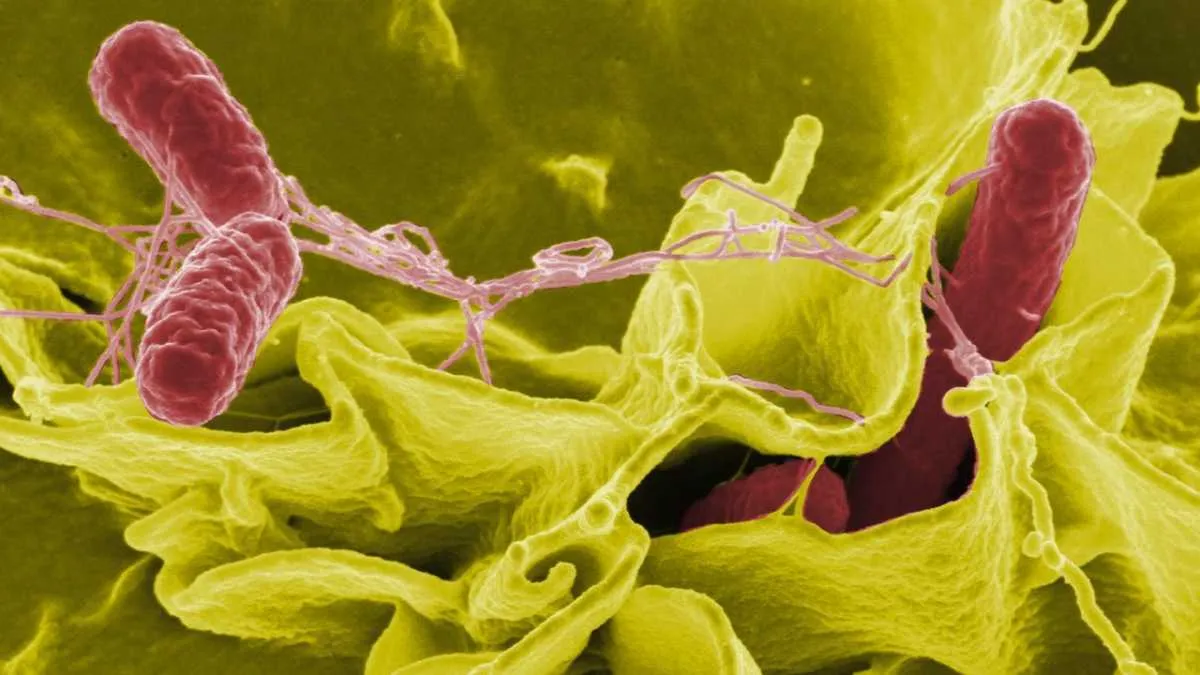
Salmonella bacteria are hugely diverse, comprising thousands of strains (serotypes) within two main species. Some are host-specific, whilst others can colonise humans and a range of animals. Humans can encounter the bacterium via contact with infected animals, but the most common route is through poorly cooked food. They cause what is collectively known as salmonellosis – essentially food poisoning, characterised by one or more of the following symptoms: fever, abdominal pain, diarrhoea, nausea and sometimes vomiting. In particularly vulnerable patients like the young and elderly, antibiotics can be necessary and some resistant serotypes of Salmonella have now emerged.
2
Helicobacter pylori
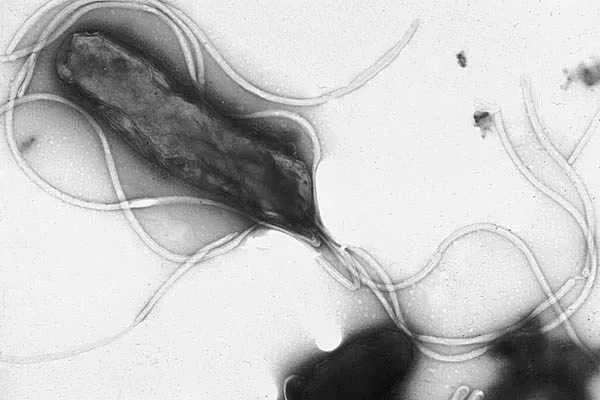
This bacterium colonises the lining of the stomach and is estimated to be present in up to two-thirds of the human population, usually without any symptoms. When it flares up and becomes a problem, it causes stomach ulcers, which usually require antibiotic treatment. Combinations of antibiotics that kill in different ways are currently being used to help counter resistance in this bacterium, which has already arisen to the antibiotic clarithromycin.
3
Enterobacteriaceae
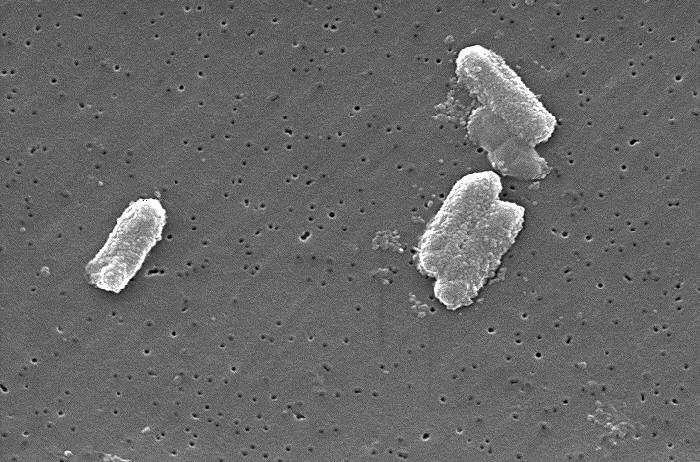
This is a large and diverse family of bacteria inhabiting the human gut. Most are harmless symbionts (your so-called ‘friendly’ bacteria) but there are some unpleasant pathogens amongst them including Salmonella, E.coli, Klebsiella and Shigella which are showing resistance to our most powerful group of antibiotics, the carbapenems. Carbapenem-resistant enterobacteriaceae, or CRE as they are known, are largely encountered in healthcare settings and can be fatal in up to 50 per cent of cases (E. coli and Klebsiella are the major bacteria causing blood-poisoning). They can now only be treated by a small and dwindling number of last-resort antibiotics, and even those are not effective in every single case. CRE are amongst the most critical targets for next-generation antibiotics. Members of the wider population are generally not at risk.
4
Campylobacter spp.
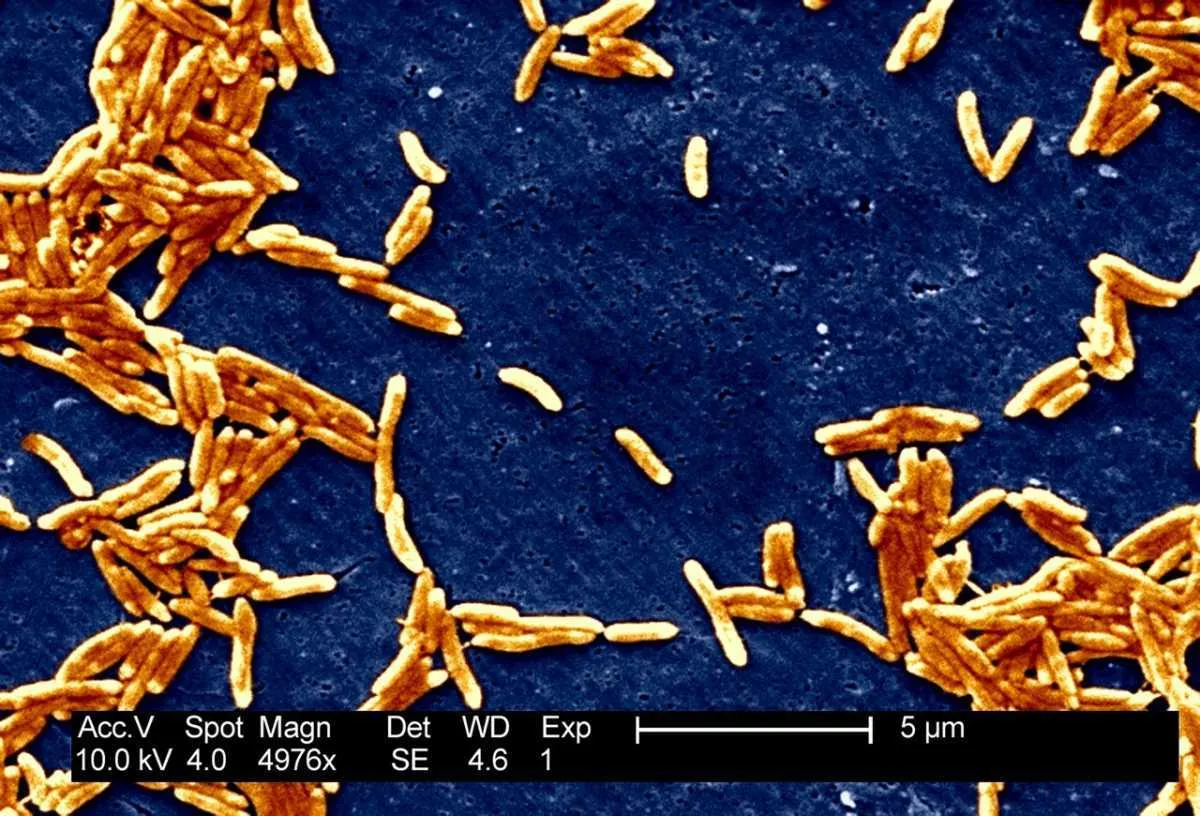
Campylobacter bacteria occur naturally in the bodies of many food animals and so the route of transmission to humans is usually through raw or under-cooked meat. The bacterium can be killed easily by heating, but once in your system it can lead to serious food-poisoning symptoms and additional complications. Treatment with antibiotics is not usually necessary, but their inappropriate use in some parts of the world has led to resistance to antibiotics such as the fluoroquinolones.
5
Neisseria Gonorrhoeae
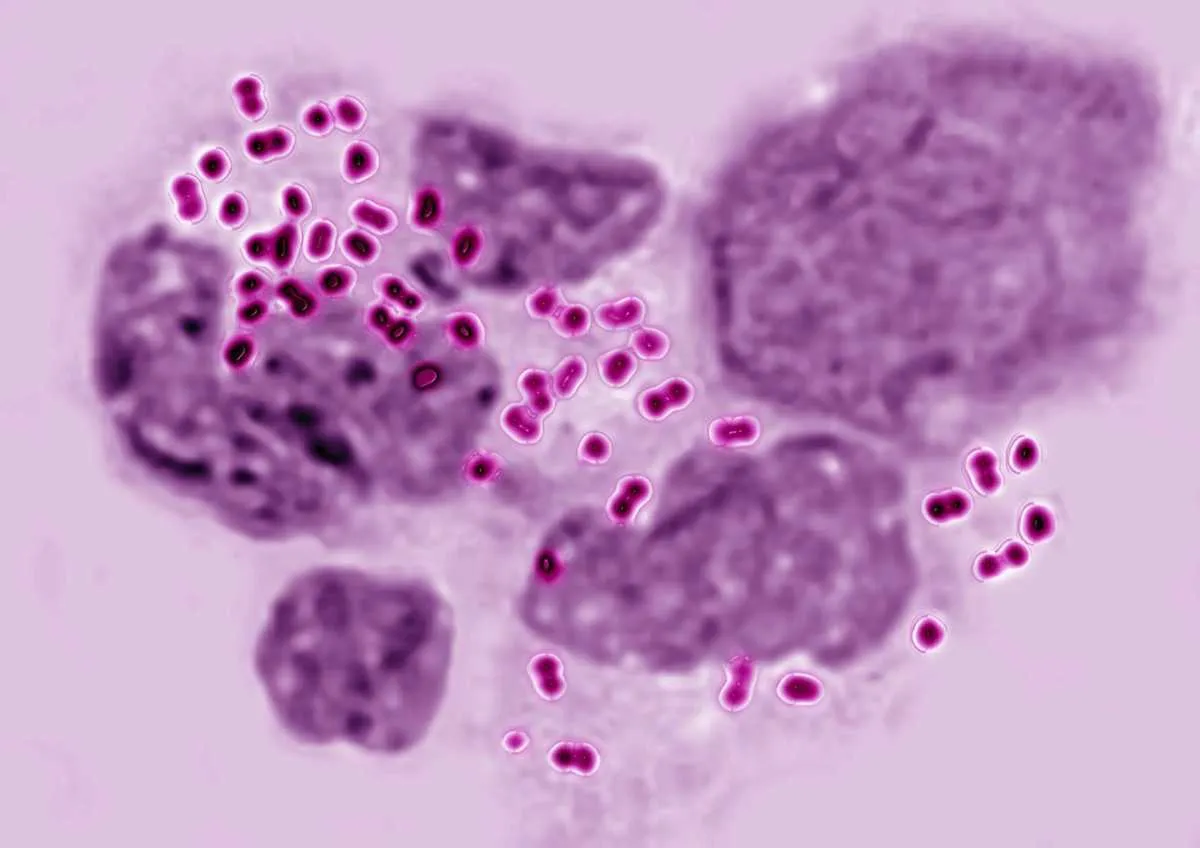
Responsible for the sexually-transmitted infection gonorrhoea, increasing antibiotic resistance has been noted since the 1940s for penicillins, tetracyclines, quinolones and macrolide antibiotics. More recently, the bacterium has shown resistance to third-generation cephalosporins and is officially regarded as “multi-drug resistant”.
6
Pseudomonas aeruginosa
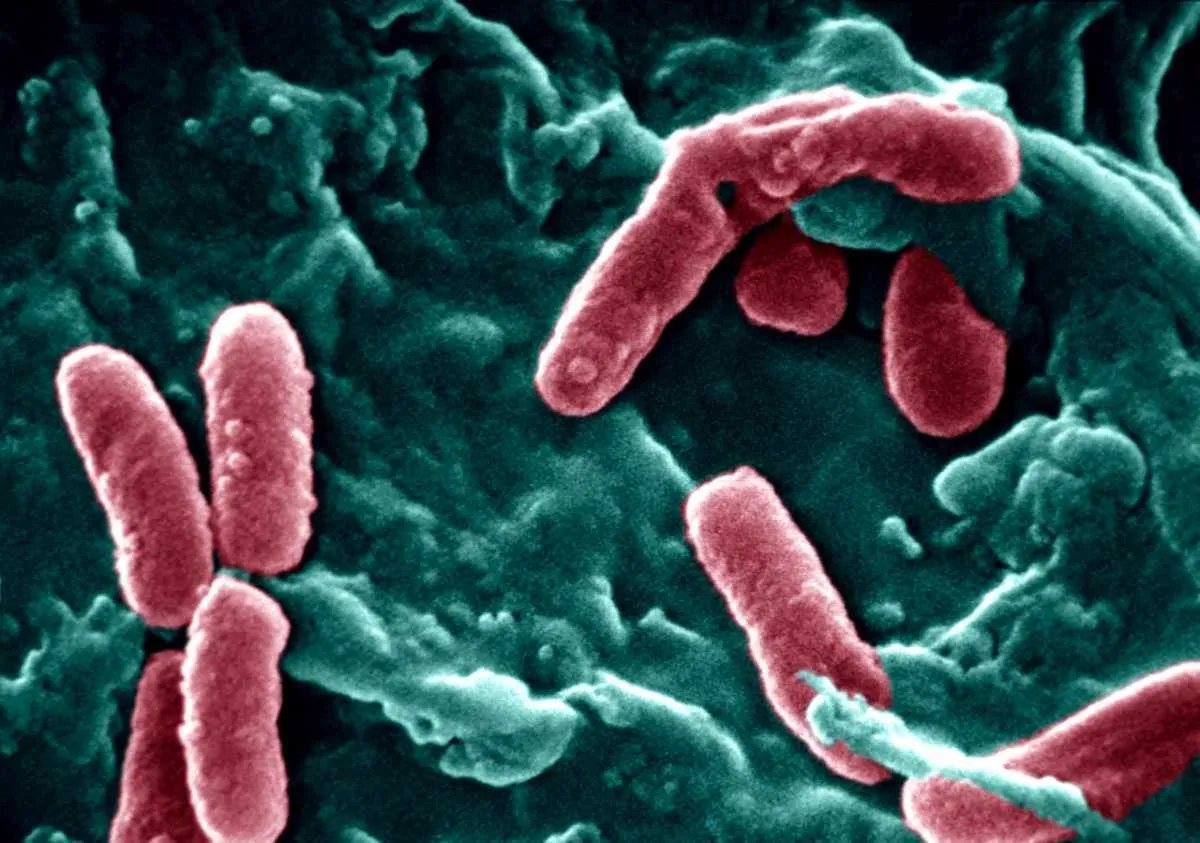
This bacterium can live in a wide range of hosts, including plants. In humans, it tends to be an opportunistic pathogen encountered mainly in healthcare settings. It is officially “multi-drug resistant” and amongst the most critical threats we face, now resistant to several ‘last resort’ antibiotics amongst the carbapenems and third-generation cephalosporins. It is feared that this bacterium could become untreatable and life-threatening in the very near future.
7
Enterococcus faecium
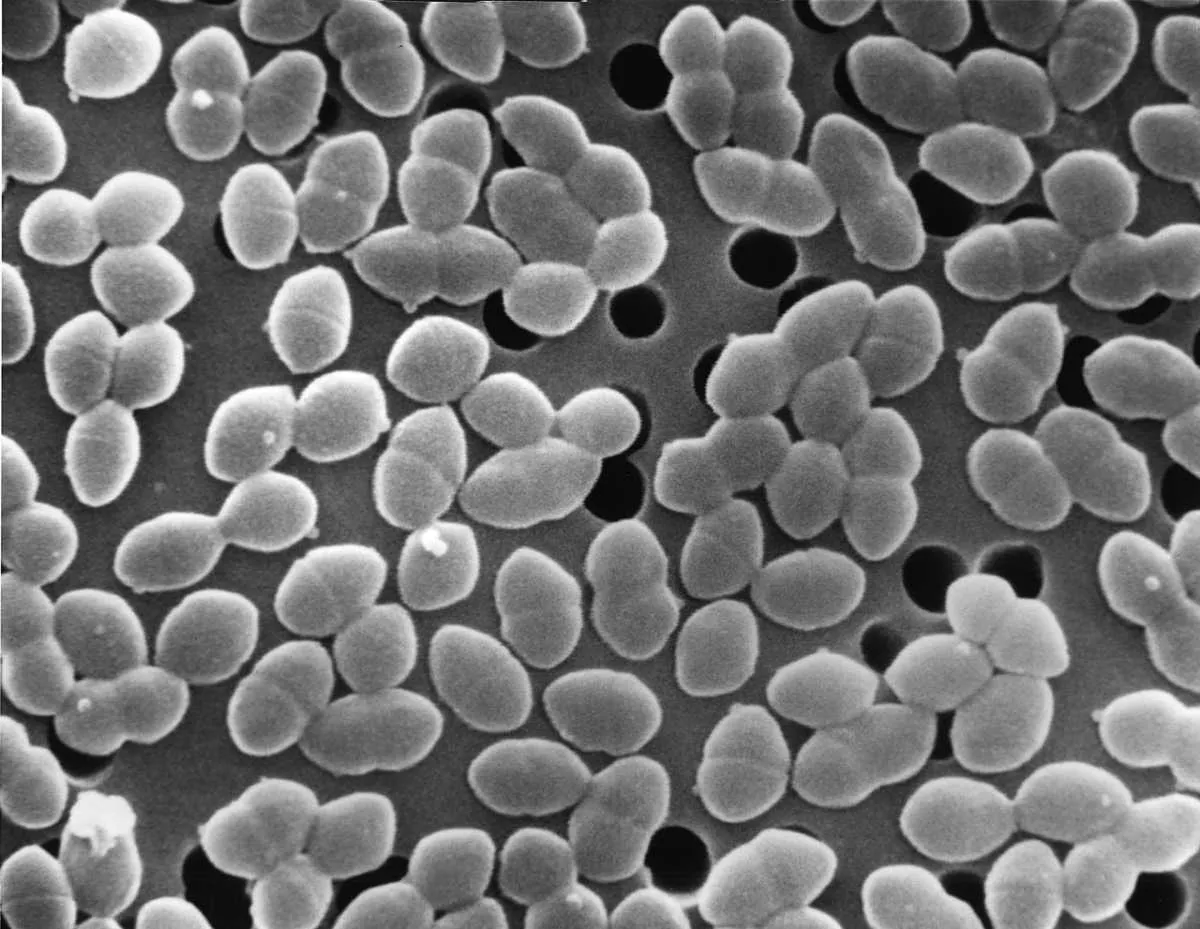
Common in the human intestine, E. faecium lives peacefully with us most of the time. However, it can turn pathogenic, causing serious illnesses such as urinary tract infections, blood infections and meningitis. It has intrinsic resistance to several classes of antibiotics, such as penicillins and cephalosporins, but is most well-known for its resistance to the antibiotic vancomycin. Certain combination therapies are now needed to defeat vancomycin-resistant enterococci, or VRE as they are known.
8
Staphylococcus Aureus (MRSA)
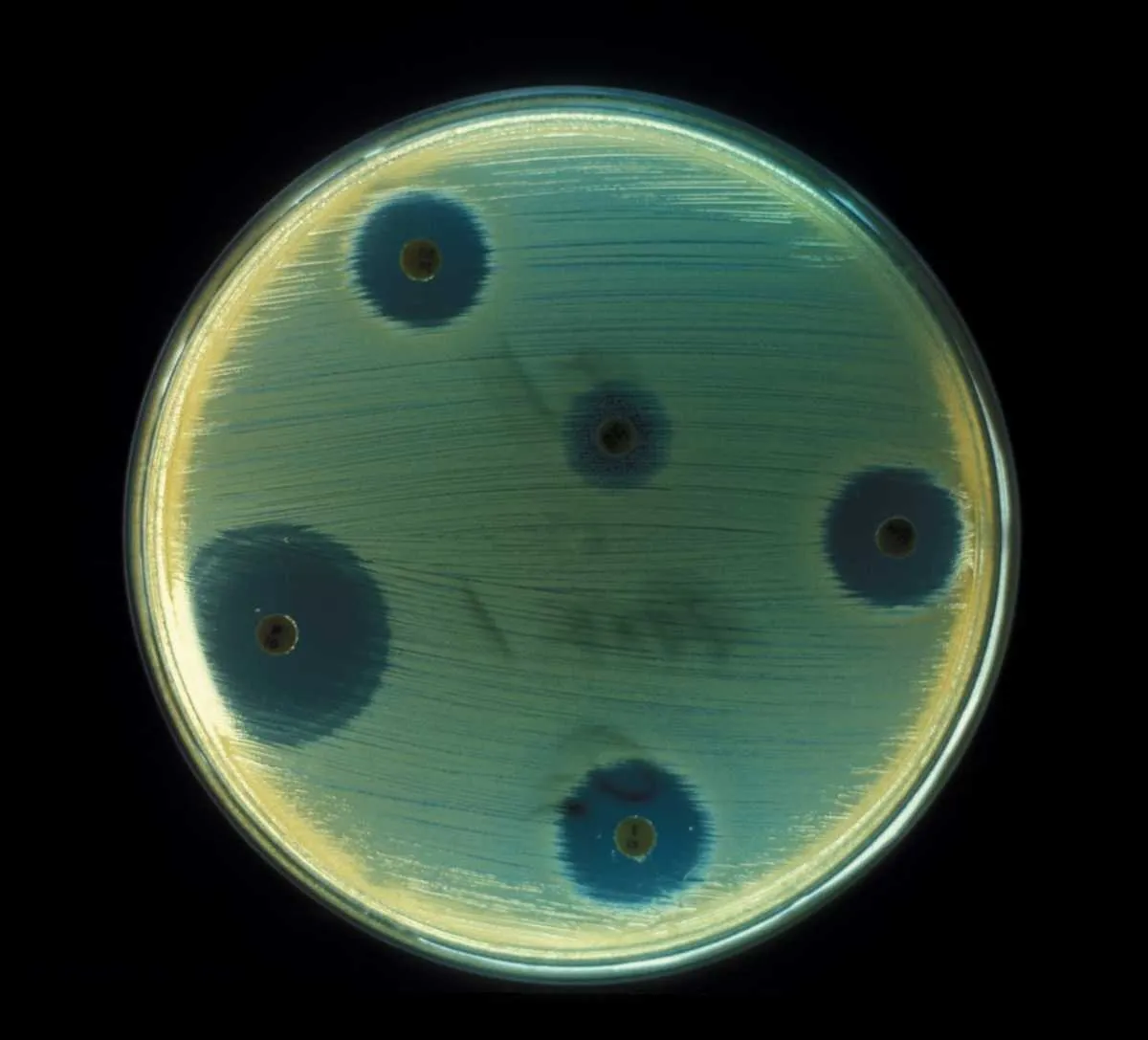
Most strains of Staph aureus (as it is known in short) live harmlessly on the skin and linings of the nose and airways. However, it can turn pathogenic if it enters deeper into the body, for example through wounds. In healthcare settings, one strain has emerged to cause serious problems – methicillin-resistant Staphylococcus aureus, or MRSA. It is the original and archetypal “superbug”, now resistant to all but a handful of our ‘last-resort’ antibiotics. It has been found in the wider community, outside of healthcare settings.
9
Acinetobacter Baumannii
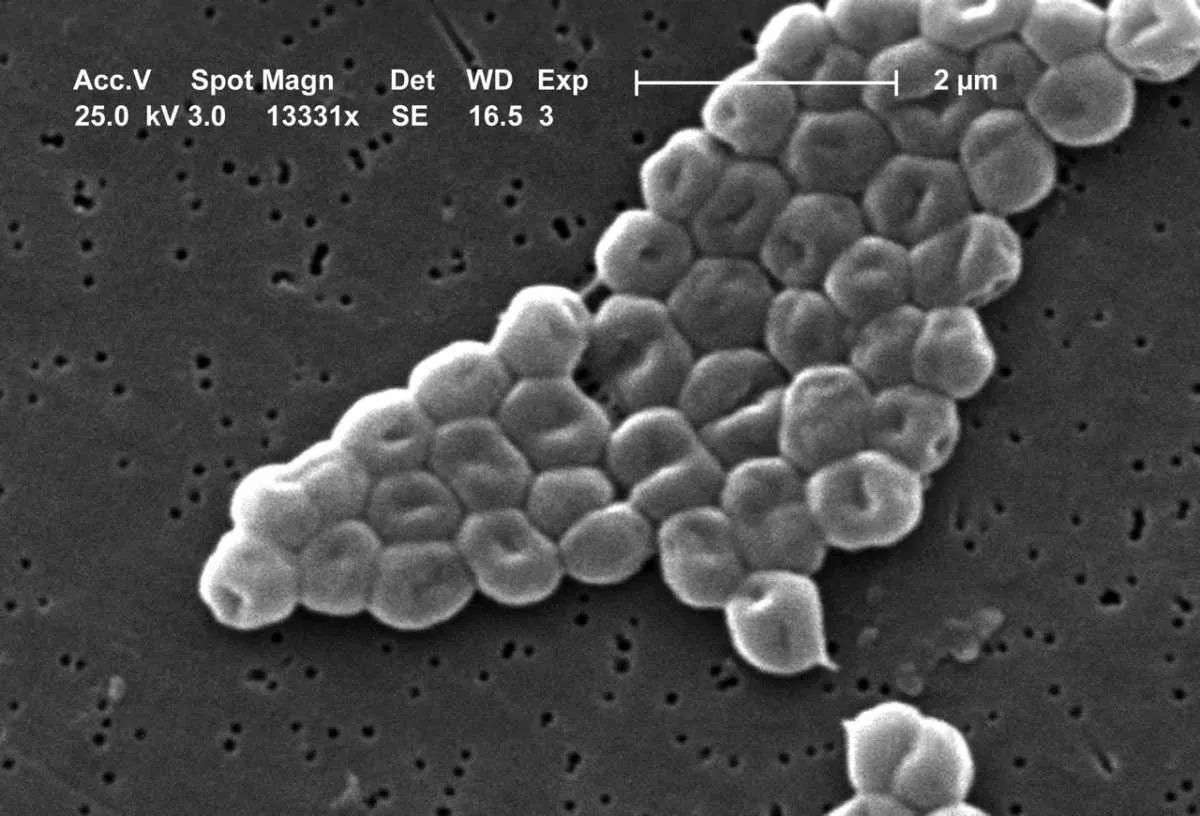
This bacterium could be regarded as ‘public enemy number one’. It’s on the critical list for next generation antibiotics, having defeated our ‘last-resort’ carbapenem antibiotics. It causes pneumonia and wound infections, which can lead to untreatable blood infections (sepsis). Occurring mainly in healthcare settings, it leads to high mortality. It has a particularly strong tendency for getting genetic material from other species of bacteria, which is why it has been so successful in developing resistance to almost everything we can throw at it.
This is an extract fromUnderstand Antibiotic Resistanceinissue 310ofBBC Focusmagazine –subscribe hereto make sure you get the full features delivered to your door before they hit the newsstands.