Human trials of two coronavirus vaccine candidates are underway in the UK.
Both teams, at the University of Oxford, and Imperial College London, hope to have results by the end of the year and have secured deals for manufacturing.
But there are also trials ongoing across the world, as researchers race to find a way to protect people from COVID-19.
The University of Oxford vaccine
Researchers at the University of Oxford announced the start of phase II and III UK clinical trials last month.
The phase I trial in healthy adult volunteers began in April. More than 1,000 immunisations have been completed and follow-up is currently ongoing.
More than 4,000 participants are already enrolled in the UK, with enrolment of a further 10,000 people planned as researchers test the vaccine – known as ChAdOx1 nCoV-19.

Trials are also taking place in South Africa and Brazil.
It is hoped results of the trial might be available by September, with an effective coronavirus vaccine being ready soon after.
ChAdOx1 nCoV-19 is made from a virus (ChAdOx1), which is a weakened version of a common cold virus (adenovirus) that causes infections in chimpanzees, that has been genetically changed so that it is impossible for it to replicate in humans.
Imperial College London vaccine
On 19 June the first healthy volunteer received a small dose of a potential COVID-19 vaccine developed by researchers from Imperial College London.
The participant is reported to be in good health with no safety concerns after being injected.
A second booster dose will follow within four weeks.
Around 300 healthy participants are expected to take part in this trial.
Imperial’s RNA vaccine uses synthetic strands of genetic code based on the genetic material of Sars-CoV-2, the coronavirus responsible for the pandemic.
It works by delivering genetic instructions to muscle cells to make the “spike” protein on the surface of Sars-CoV-2.
The presence of this protein provokes an immune response, offering protection against COVID-19.
If the vaccine is safe and shows promising immune response, a further trial involving 6,000 people is expected to go ahead in October.
Other coronavirus vaccines from around the world
In March, scientists in the US started human trials for a vaccine.
In May they said early indications were that the vaccine could train the immune system to fight coronavirus.
They reported that neutralising antibodies were found in the first eight people who took part in safety trials for the experimental mRNA-1273 vaccine.
The drug, being tested by firm Moderna, injects a small sample of COVID-19’s genetic code into patients.
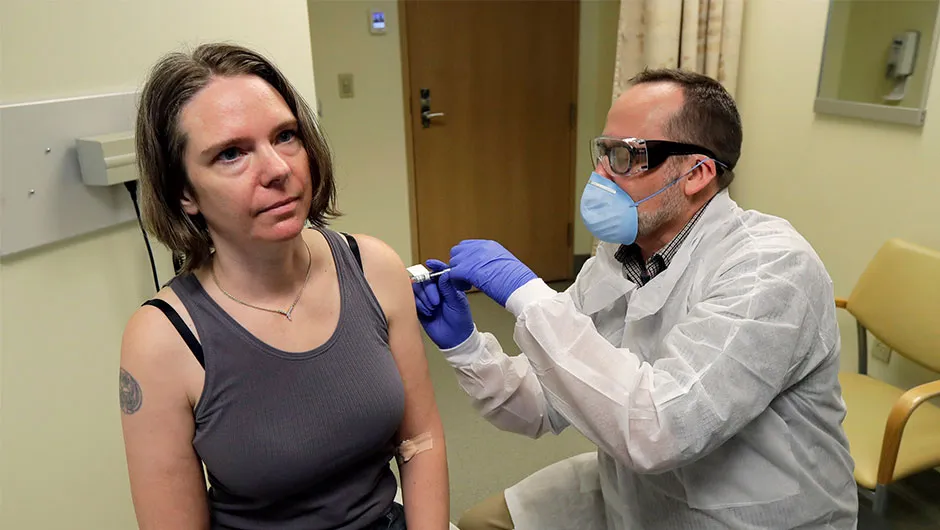
Volunteer Jennifer Haller was the first person to receive a dose of a potential vaccine for COVID-19 at a Seattle health research institute © Ted S Warren/AP
Elsewhere, pharmaceutical companies Sanofi and GSK have teamed up with the hope of making a COVID-19 vaccine available by the middle of next year.
They are hoping to enter a candidate in clinical trials in the second half of this year.
Scientists at the University of Pittsburgh School of Medicine are working on a fingertip-sized patch which could be a potential vaccine for coronavirus.
Vaccines candidates are also being trialled in a number of other countries across the world, including China and Australia.
If a successful vaccine is developed, can it be manufactured to scale?
Production of the vaccine has already been scaled up ahead of the trial to prepare as early as possible for potential future deployment.
AstraZeneca has reached an agreement with Europe’s Inclusive Vaccines Alliance (IVA), to supply up to 400 million doses of the University of Oxford’s COVID-19 vaccine – at no profit, with deliveries starting by the end of 2020.
Read the latest coronavirus news:
- Simple salt water solution 'could help reduce symptoms'
- Male donor plasma 'contains more coronavirus antibodies' than female
- COVID-19 fatalities 'very rare' in children
Meanwhile, the British government has agreed to pay for up to 100 million doses, adding that 30 million may be ready for UK citizens by September.
The company has also completed similar agreements with the UK, US, the Coalition for Epidemic Preparedness Innovations and Gavi the Vaccine Alliance for 700 million doses.
It has further agreed a licence with the Serum Institute of India for the supply of an additional one billion doses, principally for low and middle-income countries.
Its total manufacturing capacity currently stands at two billion doses.
Imperial has also formed a new social enterprise called VacEquity Global Health (VGH) to develop and distribute its vaccine across the UK and worldwide.
UK-headquartered GlaxoSmithKline has joined forces with a raft of partners around the world to develop a number of vaccines for the disease.
Other companies like Johnson and Johnson and Pfizer are also working on vaccine candidates.


