- Research develop a fingertip-sized patch of 400 tiny needles that deliver vaccine into the skin, where the immune reaction is strongest.
- The new vaccine can be mass produced and can sit at room temperature until needed, eliminating the need for refrigeration during transport or storage.
- Researchers in the US say they were able to act quickly because they had laid the groundwork during previous epidemics.
A fingertip-sized patch could be a potential vaccine for coronavirus, researchers say.
Scientists suggest that when tested in mice, it produced antibodies specific to COVID-19 [PDF] in quantities thought to be sufficient for neutralising the virus.
The vaccine is described in a paper published in EBioMedicine, which is published by The Lancet, and is thought to be the first to be reviewed by other scientists.
Researchers at the University of Pittsburgh School of Medicine say they were able to act quickly because they had laid the groundwork during earlier coronavirus epidemics.
Read the latest coronavirus news:
- Coronavirus 'most infectious' in first weeks of symptoms
- Social distancing measures working, study suggests
- Behavioural scientists 'very concerned' about mass coronavirus testing
Co-senior author Andrea Gambotto, associate professor of surgery at the university, said: “We had previous experience on SARS-CoV in 2003 and Mers-CoV in 2014.
“These two viruses, which are closely related to SARS-CoV-2, teach us that a particular protein, called a spike protein, is important for inducing immunity against the virus. We knew exactly where to fight this new virus.
“That’s why it’s important to fund vaccine research. You never know where the next pandemic will come from.”
The researchers say their vaccine candidate follows a more established approach compared to the experimental mRNA vaccine candidate that has entered clinical trials in Seattle.
They have called their drug PittCoVacc, short for Pittsburgh Coronavirus Vaccine, and it uses lab-made pieces of viral protein to build immunity – the same way that current flu jabs work.
The researchers also used a novel approach to deliver the drug, called a microneedle array, to increase potency.
This array is a fingertip-sized patch of 400 tiny needles that delivers the spike protein pieces into the skin, where the immune reaction is strongest.
The patch is applied like a plaster and then the needles, which are made entirely of sugar and the protein pieces, simply dissolve into the skin.
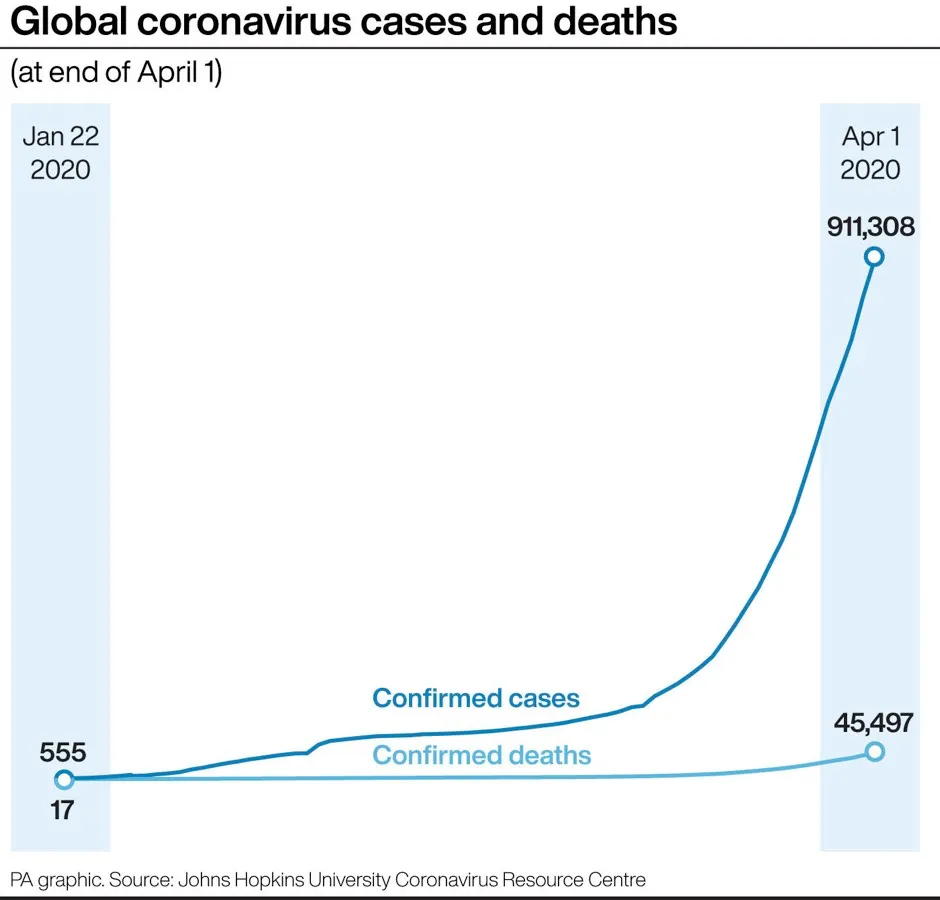
Co-senior author Louis Falo, professor and chair of dermatology at the School of Medicine and University of Pittsburgh Medical Centre, said: “We developed this to build on the original scratch method used to deliver the smallpox vaccine to the skin, but as a high-tech version that is more efficient and reproducible patient to patient.
“And it’s actually pretty painless – it feels kind of like Velcro.”
Discover more about the coronavirus:
- Corrupted Blood: what the virus that took down World of Warcraft can tell us about coronavirus
- Will COVID-19 become a seasonal virus?
- Coronavirus: can herd immunity protect us from COVID-19?
- Is hand-washing really the best thing we can do to stop the spread of COVID-19?
Researchers say their system can be mass produced, and once manufactured, the vaccine can sit at room temperature until it is needed, eliminating the need for refrigeration during transport or storage.
According to the study, when tested in mice, PittCoVacc generated a surge of antibodies against SARS-CoV-2 within two weeks of the microneedle prick.
The authors are now in the process of applying for an investigational new drug approval from the US Food and Drug Administration in anticipation of starting a phase I human clinical trial in the next few months.
How do scientists develop vaccines for new viruses?
Vaccines work by fooling our bodies into thinking that we’ve been infected by a virus. Our body mounts an immune response, and builds a memory of that virus which will enable us to fight it in the future.
Viruses and the immune system interact in complex ways, so there are many different approaches to developing an effective vaccine. The two most common types are inactivated vaccines (which use harmless viruses that have been ‘killed’, but which still activate the immune system), and attenuated vaccines (which use live viruses that have been modified so that they trigger an immune response without causing us harm).
A more recent development is recombinant vaccines, which involve genetically engineering a less harmful virus so that it includes a small part of the target virus. Our body launches an immune response to the carrier virus, but also to the target virus.
Over the past few years, this approach has been used to develop a vaccine (called rVSV-ZEBOV) against the Ebola virus. It consists of a vesicular stomatitis animal virus (which causes flu-like symptoms in humans), engineered to have an outer protein of the Zaire strain of Ebola.
Vaccines go through a huge amount of testing to check that they are safe and effective, whether there are any side effects, and what dosage levels are suitable. It usually takes years before a vaccine is commercially available.
Sometimes this is too long, and the new Ebola vaccine is being administered under ‘compassionate use’ terms: it has yet to complete all its formal testing and paperwork, but has been shown to be safe and effective. Something similar may be possible if one of the many groups around the world working on a vaccine for the new strain of coronavirus (SARS-CoV-2) is successful.
Read more:


